A Randomized Controlled Trial Evaluating the Efficacy of E-Cigarette Use for Smoking Cessation in the General Population: E3 Trial Design
Background: Smoking cessation improves morbidity and mortality among smokers who achieve long-term abstinence. Many smokers are using electronic cigarettes (e-cigarettes) to attempt to quit, despite a lack of data concerning their efficacy and safety for smoking cessation.
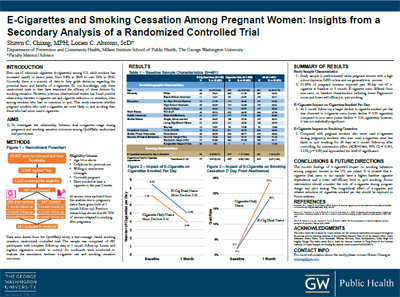
E-Cigarettes and Smoking Cessation Among Pregnant Women: Insights From a Secondary Analysis of a Randomized Controlled Trial
Background: The use of electronic cigarettes is a rapidly expanding phenomenon. Currently, there is a scarcity of data to help guide decisions regarding the potential harm and benefits of e-cigarettes. This study examines whether pregnant smokers who used e-cigarettes are more likely to quit smoking than those who had never used e-cigarettes.
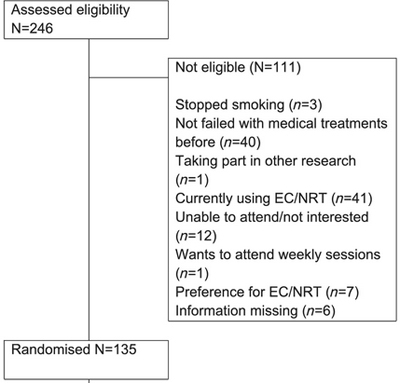
E-cigarettes versus nicotine replacement treatment as harm reduction interventions for smokers who find quitting difficult: randomized controlled trial
The majority of smokers accessing the current best treatments continue to smoke. We aimed to test if e-cigarettes (EC) compared with nicotine replacement treatment (NRT) can help such smokers to reduce smoking.
EffiCiency and Safety of an eLectronic cigAreTte (ECLAT) as Tobacco Cigarettes Substitute: A Prospective 12-Month Randomized Control Design Study
Electronic cigarettes (e-cigarettes) are becoming increasingly popular with smokers worldwide. Users report buying them to help quit smoking, to reduce cigarette consumption, to relieve tobacco withdrawal symptoms, and to continue having a 'smoking' experience, but with reduced health risks. Research on e-cigarettes is urgently needed in order to ensure that the decisions of regulators, healthcare providers and consumers are based on science.
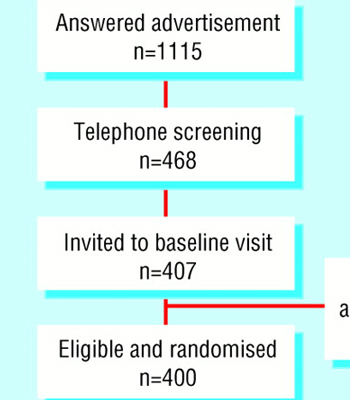
Electronic cigarettes for smoking cessation: a randomised controlled trial
Electronic cigarettes (e-cigarettes) can deliver nicotine and mitigate tobacco withdrawal and are used by many smokers to assist quit attempts. We investigated whether e-cigarettes are more effective than nicotine patches at helping smokers to quit.
Smoking reduction with oral nicotine inhalers: double blind, randomised clinical trial of efficacy and safety
Objectives: To determine whether use of an oral nicotine inhaler can result in long term reduction in smoking and whether concomitant use of nicotine replacement and smoking is safe.
Design: Double blind, randomised, placebo controlled trial. Four month trial with a two year follow up.
E-cigarettes May Support Smokers With High Smoking-Related Risk Awareness to Stop Smoking in the Short Run: Preliminary Results by Randomized Controlled Trial
E-cigarettes may be positively used in tobacco cessation treatments. However, neither the World Health Organization nor the American Food and Drug Administration has recognized them as effective cessation aids. Data about the efficacy and safety of e-cigarettes are still limited and controversial.
A Randomized Trial Comparing the Effect of Nicotine Versus Placebo Electronic Cigarettes on Smoking Reduction Among Young Adult Smokers
Electronic cigarette (EC) use is growing dramatically with use highest among young adults and current smokers. One of the most common reasons for using ECs is interest in quitting or reducing cigarettes per day (CPD); however there are few randomized controlled trials (RCT) on the effect of ECs on smoking abstinence and reduction.
Research
Reports
Featured Posts
EffiCiency and Safety of an eLectronic cigAreTte (ECLAT) as Tobacco Cigarettes Substitute: A Prospective 12-Month Randomized Control Design Study
 as Tobacco Cigarettes Substitute A Prospective 12-Month Randomized Control Design Study.png)